Zinc-Iron
-
Thread to keep track of our zinc-iron develepment. This is quite preliminary, based roughly off of Savinell's work.
Zinc is preferentially plated on the negative side, even though the potential for doing so is more negative than iron, which is present in solution.
SOC ranges tested have been quite minimal however.
Some of @danielfp@chemisting.com's initial testing is here:
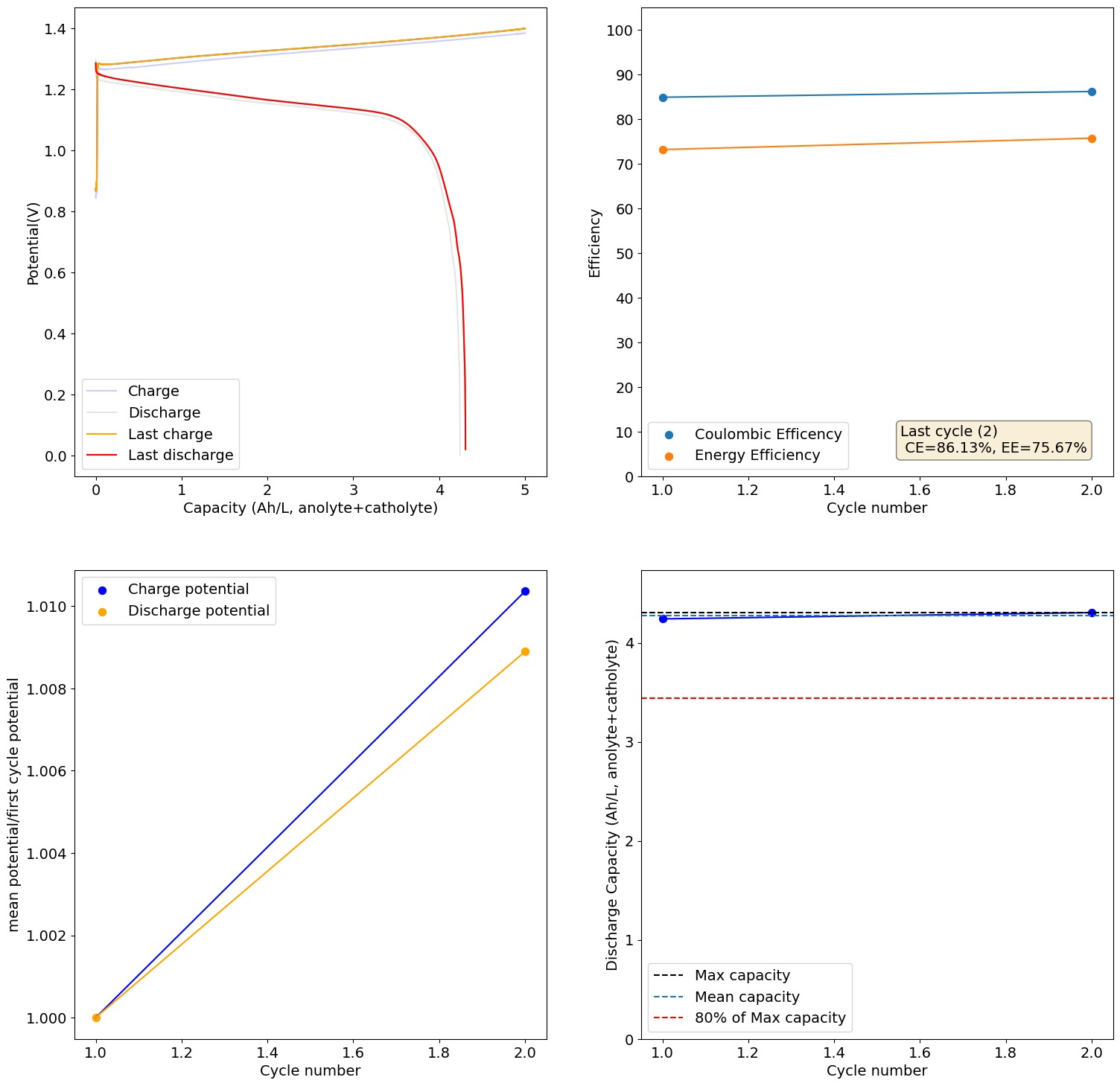
From Daniel:
This is 2M FeCl2, 3M ZnCl2, 2M Glycine. At 20mA/cm2. Felt on both sides, daramic membrane. The pH of this is 3.2, but the CE is quite high so H2 generation must be quite low.
Running to higher capacities you get dendrites quite quickly. I am trying 2M FeCl2, 3M ZnCl2, 2M Glycine with 1M NH4Cl with the nonconductive felt on the anode.
I'm also going to change to the fancy pumps with PTFE tubing after this run.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login