Fe-Mn
-
@danielfp248 I ran into some of your old work on Fe-Mn batteries. I've been interested in Fe-Mn batteries for some time and was wondering if you could share some of your experience.
In particular, if we look at the pourbaix diagram of Fe and Mn overlapped, there's a region in the pH 4-6 range where Mn2+ oxidizes to MnO2 and Fe2+ reduces to Fe on charge.
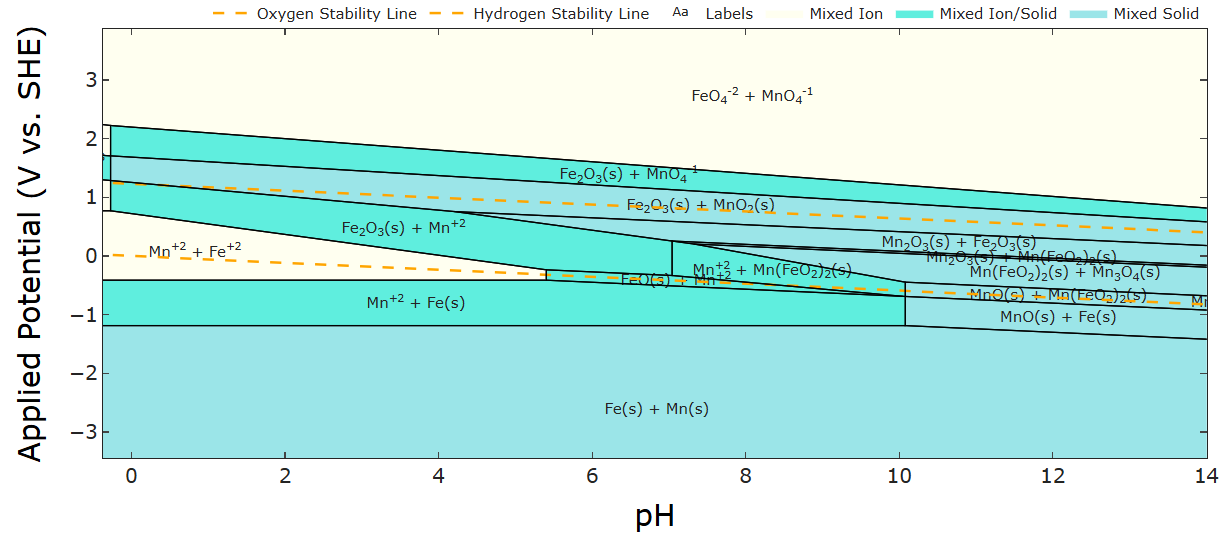
Let's say we can ignore the fact that there are solid phases - then ion crossover poses a long term concern. Have you guys found any inexpensive or DIY ion selective membrane options (either specific to a particular ion or broad spectrum diy anion/cation exchange membranes?) In my experience screening for inexpensive battery chemistries, crossover of solution phase species is a problem I haven't really seen an easy DIY solution for. Not that I've looked super hard!
-
@danielfp248 I ran into some of your old work on Fe-Mn batteries. I've been interested in Fe-Mn batteries for some time and was wondering if you could share some of your experience.
In particular, if we look at the pourbaix diagram of Fe and Mn overlapped, there's a region in the pH 4-6 range where Mn2+ oxidizes to MnO2 and Fe2+ reduces to Fe on charge.

Let's say we can ignore the fact that there are solid phases - then ion crossover poses a long term concern. Have you guys found any inexpensive or DIY ion selective membrane options (either specific to a particular ion or broad spectrum diy anion/cation exchange membranes?) In my experience screening for inexpensive battery chemistries, crossover of solution phase species is a problem I haven't really seen an easy DIY solution for. Not that I've looked super hard!
@muntasirms I've done several experiments on Fe/Mn and Zn/Mn chemistries. The problem with Mn2+ is the formation of the solid MnO2 phase and the presence of the metastable Mn3+. Forming solid MnO2 poses a non-trivial constraint on the battery, as it limits deposition per area to around a few mAh/cm2, very impractical for a flow battery, furthermore, Mn3+ formation causes MnO2 to form away from the electrode (as it disproportionates into Mn2+ and MnO2), causing some Mn to become lost around the battery system.
A possibility is to try to stabilize Mn3+ somehow (for example with Mn-EDTA), but the main issue is that even this stabilized Mn3+ is unstable and eventually self-degrades by oxidizing the chelate around the Mn atom. I tried creating a flow battery system with Fe-DTPA/Mn-EDTA, which has a max solubility of around 0.5M, but the system did not cycle due to the Mn-EDTA being too unstable. There are a few posts on my blog about this. The oxidized Mn-EDTA is also quite sensitive to pH, so it is hard to create conditions under which it is stable. Mn3+ can also be stabilized with HCl+H2SO4, but only at very low concentrations (there's a paper on using this in a flow battery, but only very low capacities are achieved).
Another possibility is to stabilize MnO2 as nanoparticles in solution. This can be achieved through the use of TiO2+ in sulfuric acid (using titanyl sulfate). Such systems are quite harsh from a chemical perspective, so I haven't tested them at all (I don't want to run a 3M sulfuric acid system containing reactive Ti compounds). You can read more about this system here (https://www.sciencedirect.com/science/article/pii/S0378775322000209). This is one of the most interesting and potentially viable Mn chemistries out there although only reaching around 17Wh/L.
Honestly Mn based systems are best suited for static batteries, where the formation of the MnO2 and Mn3+ phases is less problematic.
-
I was doing a literature review of Fe/Mn the other day and happened to find this article on Fe/Mn using MSA (https://www.sciencedirect.com/science/article/pii/S001346862030637X). This article uses an asymmetric setup with FeCl3 on one side (paper says it's FeCl2 but that must be a mistake because the reaction requires Fe reduction on charge) and MnCl2 on the other, both sides using 3M methanesulfonic acid, separated by a Nafion membrane. The Mn3+ is in theory stabilized in the acid media, but given the color of the solution it might be that MnO2 nanoparticles are stabilized instead.
While the paper does not use this in a symmetric setup, I see no reason why this reaction couldn't work symmetrically so I prepared an electrolyte using the following:
- 3g MnCl2.4H2O
- 4mL FeCl3 40% w/w solution
- 4mL 75% methanesulfonic acid (MSA)
- around 1mL of water (final volume was taken to 10mL)
The above creates a solution that is around 1.5M Fe, 1.5M Mn and 3M MSA. This setup has the advantage that both reactions generate no solid products. At a 100% SOC this would give us ~20Ah/L. On charge:
Fe3+ + e- -> Fe2+
Mn2+ -> Mn3+ + e-The potential difference between these two half reactions is not very high though, so the total expected cell voltage is ~550mV. However this is a "true flow battery" in that power and capacity are fully decoupled as the reaction products are all in solution. Note that Mn3+ is expected to have limited stability, especially at high concentrations, so I would expect capacity to degrade heavily as the Mn3+ gets converted into MnO2, unless this MnO2 is somehow stabilized in solution (which could be as nanoparticles). Interestingly Fe2+ can react with MnO2, so the battery might also self-heal if this happens, just temporarily capacity in the process.
I loaded the electrolyte in a cell with carbon felt on both anode and cathode and used Daramic as a separator (cannot use paper as it reacts with Mn3+). Below are the results of a few cycles at low capacity (0.25Ah/L at 10mA/cm2), just to test the chemistry. It seems to work quite well:
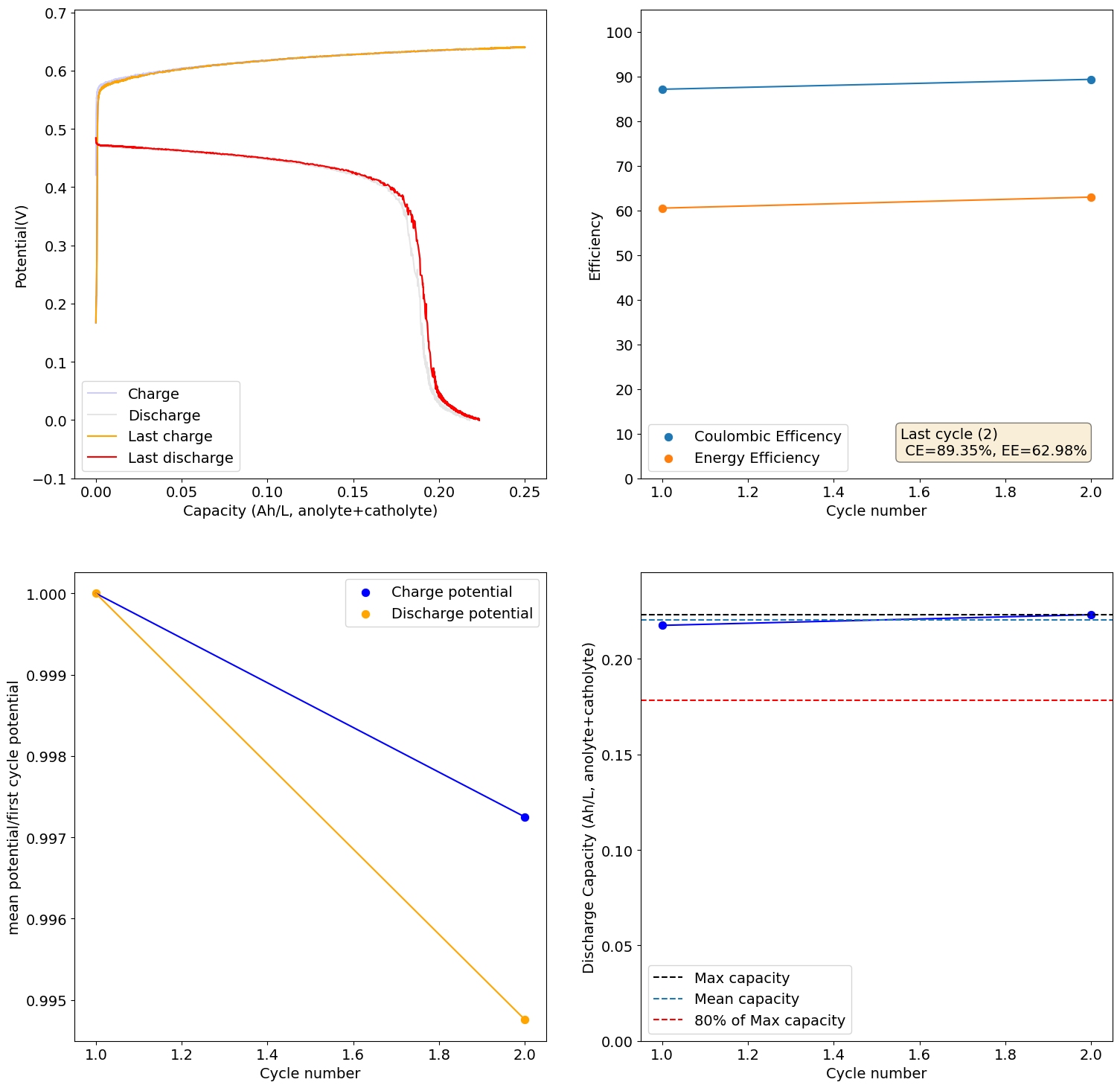
I will continue to run some tests and will let you know what I get.
-
The CE drops a lot when going to higher capacities (even at a current of 40mA/cm2, which means it is not due to crossover as lower currents do not imply a lower CE). This is likely because the stability of Mn3+ species in solution is very limited, so you seemingly cannot exceed a ~2.5Ah/L capacity.
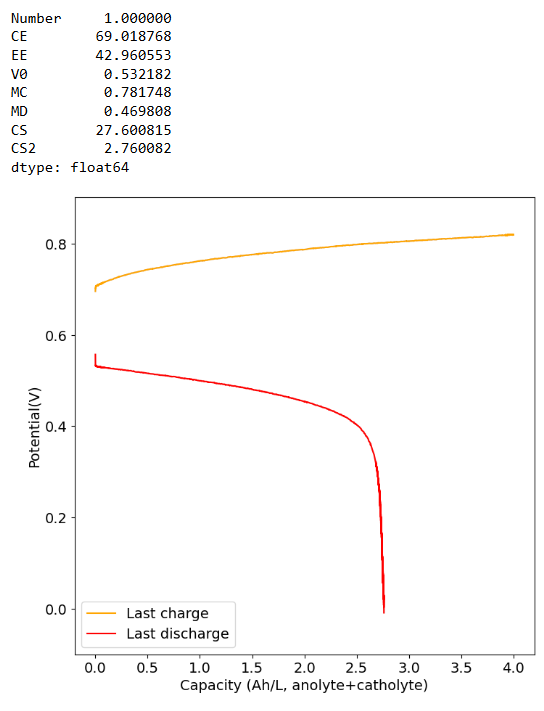
-
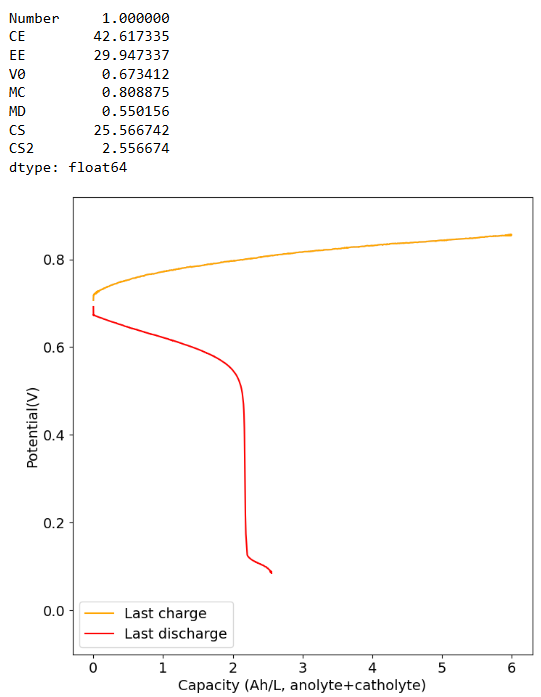
Charging to 6Ah/L at 30mA/cm2 and discharging at 5mA/cm2. At most we only get 2-3 Ah/L of available capacity, same as if we charged to 4Ah/L.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login