Made some Linux AppImages of improved MYSTAT software for anyone who uses it! Also runs on Windows ofc, it's just Python.
kirk
Posts
-
MYSTAT software v1.2.1 -
New video of cell testing processFilmed myself building a cell so you can see the whole process start-to-finish! Still working on updating the documentation for the clampable-cell branch. It works pretty darn well without any bolts.
-
Zinc-IodideIt's the same electrolyte formulation as currently in the docs
-
Zinc-Iodide -
Zinc-IodideDoing some tests now on this to get my wet lab back in action, here are the conditions and results so far on my blog
-
No pump flow battery, likely not useful, but interestingA great demo of all the transport phenomena at play in electrochemical systems!
-
Build-a-Batt - A fully parameterized flow cell stack modelAlso, this reminds me of the OpenAFPM project - they also use FreeCAD and provide a dashboard for people to input custom parameters for a small wind turbine generator, there is some FEM, and then design files are output. There is a video demo here: https://www.openafpm.net/cad-visualization
I know a few of the folks behind that project, I'm sure they'd be happy to give input on how to accomplish a similar goal but for flow batteries.
-
Build-a-Batt - A fully parameterized flow cell stack model@muntasirms Muntasir, this is awesome! Sorry for the delay in getting back to you, I've just watched your walkthrough video today. The webapp loads for me but the viewport is empty? At least on a few different browsers it seems.

I watched the walkthrough though, very nice work! It looks very clean, would be awesome actually to have a 3D interface like this on the FBRC homepage, to make it a bit cooler like the https://www.open-quantum.org project.
It would be great to share the code as open-source as well, I am not familiar with the build123d library and would like to see how you've done it.
I have also been working on some automated exports from FreeCAD, to enable some CI/CD on our documentation, but nothing web-facing: https://codeberg.org/FBRC/RFB-dev-kit/src/branch/clampable-cell/CAD/FreeCAD/FreeCAD-exports.py
@muntasirms said in Build-a-Batt - A fully parameterized flow cell stack model:
That makes boundary condition declaration and the like much easier to automate as well - so I'm in the process of slotting this into an automated hydraulics/electrochemical simulation suite. Imagine generating your custom model and simulating a "digital twin" for estimates of performance prior to purchasing materials or finalizing design.
The FreeCAD models are also fully parameterizable, and can at least automatically update in the CfdOf workbench, but the whole thing in Python would be easier to handle for hardcore simulation, with any echem phenomena. This would be really cool, I would love to see you tackle some simulation/flow frame optimization! I have my hands full currently so I'm not sure when I'd get around to it.
 Screenshot of the flow-frame file showing the OpenFoam CFD setup.
Screenshot of the flow-frame file showing the OpenFoam CFD setup.FYI, we still haven't actually done cycling experiments with the "large-format" 175 cm2 cell, so the design is mostly likely going to change somewhat!
I am not sure the small cell (18 cm2) design you show would seal also - we did a lot of testing with that sort of approach early on and it was hard to print the chemical resistant barbed fittings and endplates in one piece, which is why we separated it out in the dev kit.
@muntasirms said in Build-a-Batt - A fully parameterized flow cell stack model:
or a "fat stack" (3500cm^2) with extra, thicker bolts
I love it! I really hope we can get here one day
 we will certainly need all hands on deck to do so.
we will certainly need all hands on deck to do so.@muntasirms said in Build-a-Batt - A fully parameterized flow cell stack model:
I'm very open to constructive criticism on any part of the project (from ideation to UX to engineering). Have fun fiddling with it and let me know what you think!
Really happy that you've kicked this off, it shows whats possible in this space. Right now, I will be sticking to FreeCAD for prototyping and derisking - I'm much faster in a graphical CAD environment implementing design changes than in a code-based CAD approach. But I think the two are complementary - especially for simulation and customization.
There was actually a project that allowed for in-browser editing of FreeCAD files in a way sort of similar to your web app - a startup called Ondsel (now out of business) that made some contributions to FreeCAD source. They had a cloud tool called Lens that allowed you to build this sort of webapp directly from the FreeCAD file by exposing certain parameters. When they shut down they gave everything to FreeCAD, it looks like the Lens project is still going and you can host it yourself: https://freecad.github.io/lens-docs/
This might be a way to combine your customization approach directly with the FreeCAD source files? I would be a fan of this approach if we could link everything as closely as possible.
For UX: would be great to have a dropdown or similar to select templates for different standard configs, like the small cell/FBRC large-format/fat stack, etc.
-
Calcium-ion BatteryThis video seems to be AI generated and doesn't have that much real information? I have seen Ca-ion systems from (I think) a German group that looked good, but had a lot of organic synthetic steps if I remember correctly.
-
Using cnc router cut PVC foam boards as cell frame@rowow said in Using cnc router cut PVC foam boards as cell frame:
The long term goal and my personal next goal is to get into injection molding which of course is the best option.
At scale, definitely!
@rowow said in Using cnc router cut PVC foam boards as cell frame:
Ill see about uploading the cell fusion 360 model
A
.stepfile would also be great so I could look at it in FreeCAD! I don't have a Fusion 360 license. I'd be curious to see if we could take a similar approach. -
You Tuber Video on an open source all Iron batteryNice find! Peter Allen has been at it for a while. I'd read his previous papers but not this one. A quick scan shows they are using an ion-exchange membrane, which @danielfp248 and I try to avoid if we can, since they are often the weak point of systems that rely on them.
Crossover of the mediators through the membrane will degrade performance. The possibility of this unwanted feature was examined by cyclic voltammetry on an electrochemical cell divided by the Fumasep FAS-50 anion exchange membrane in the 2 M KCl 20 % (v/v) EG AIB 3.0 electrolyte. The initial concentrations were 10 mM MV2+ in the left chamber and 10 mM ABTS2- in the right. A constant potential of 1.25 V was applied across the cell with 6.25 cm2 graphite electrodes for 24 h. This simulates the AIB 3.0 conditions. After the 24 h period CV was performed on catholyte to evaluate MV crossover and vice versa. We found no crossover through the membrane by the cyclic Voltammetry experiments.
I like how everything is commercially available at scale. There are quite a few organic flow battery startups that have tried to use methyl viologen as well like they do in this paper. I am curious what @danielfp248 thinks
-
Upcoming improvements to the dev kit@doho said in Upcoming improvements to the dev kit:
I have try-ed to download Your new files from github (.stl and .pdf), but the files all appeared as broken, even in github.
The files to sprint for the new endplates are here: https://codeberg.org/FBRC/RFB-dev-kit/src/branch/clampable-cell/CAD/exports
I haven't updated the new
You have to download them individually, unless you clone the repository with Git LFS set up---the large CAD files are handled with LFS and without LFS installed downloading the repository just downlaods a pointer/reference to the CAD file, not the actual file (at least that's how I understand it).
Let me know if this works for you!
-
My Suction Luer LockVery nice work @doho ! It's great to see your setup.
@doho said in My Suction Luer Lock:
But for getting reproducible resultants the flow through the cell should be down to up, but not horizontal (how much would be air isolating in the chamber when changing parts?)
This is a good point, from what I've seen in other applications cells should generally clear air/produced gases by flowing against gravity from bottom to top, that is still possible with the new setup but the tubing from the pump outlet to cell inlet would have to be slightly longer. In my jig redesign I'll take this into account when writing the documentation.
-
Upcoming improvements to the dev kitAlso, we have a "double-reservoir" part that contains an internal spillover passage for systems that have a lot of water/liquid transfer. Probably going to make a version with and without this and have it as the standard going forward, to simplify the tubing and jig setup for whatever version people want to use.
The double-reservoir with spillover is in use here
-
Upcoming improvements to the dev kitOh, and we'll be trying to add Luer Lok twist connectors to make detaching the cell quickly from the rest of the setup faster/easier than undoing tight barbed fitting connections.
-
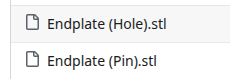
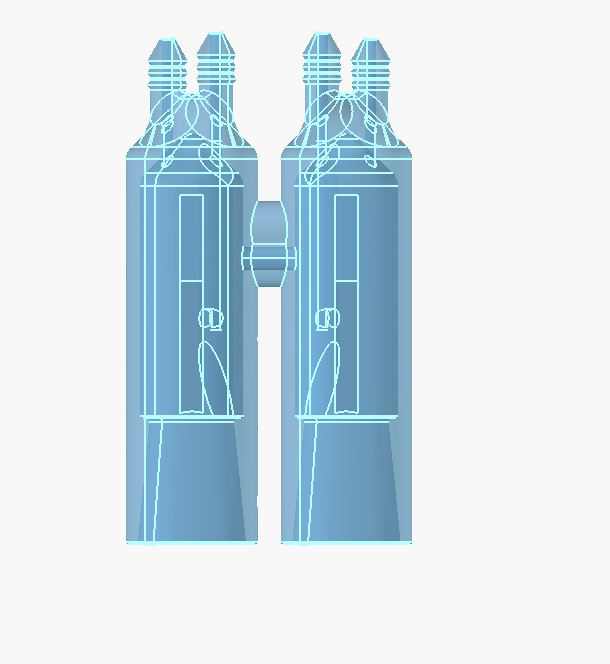
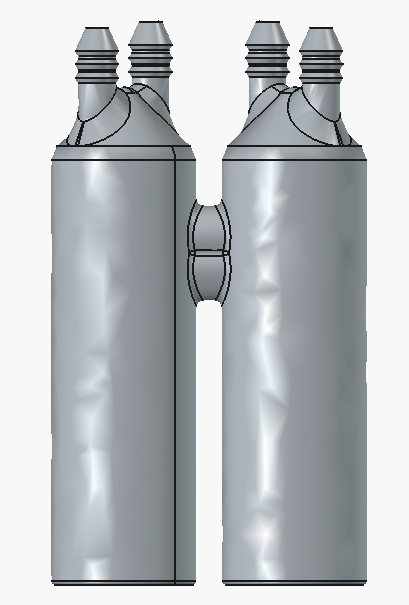
Upcoming improvements to the dev kit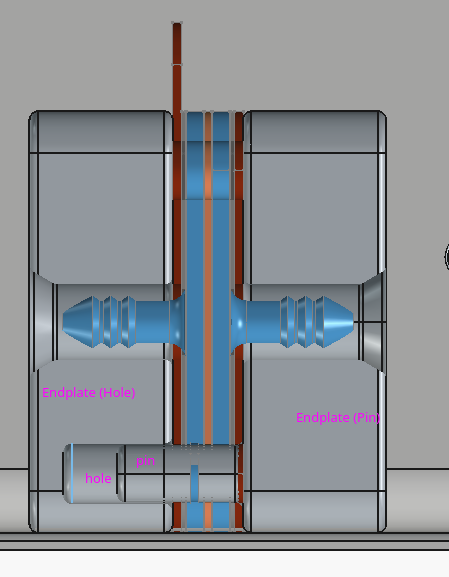

The new endplates are basically the same, just two alignment pins instead of four bolt holes.
-
Upcoming improvements to the dev kitHi all, been planning some improvements to the dev kit that are underway, based on some ideas exchanged at a conference with some other open-source flow battery projects (Redoxino and the team at QUB).
It's mostly to improve ergonomics - there is now a case to hold the arduino and wires on the rear of the cell:
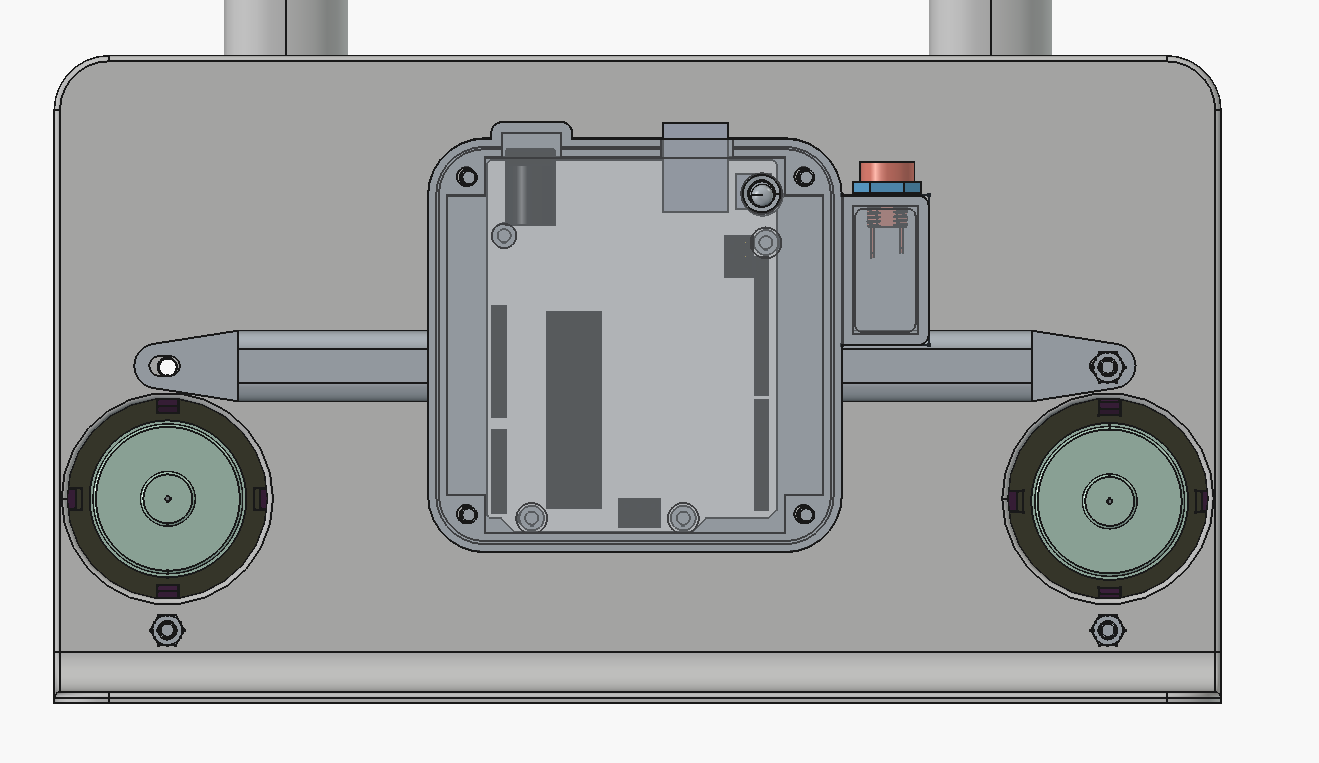
We also realized you can just clamp the cell shut with a 2-inch C-clamp instead of using the bolts, and it seals well:

Note, the new endplates here used are backwards-compatible with previous gaskets/graphite plates/brass current collectors, so no need to recut anything. Moving forward though, the design files for those components will change to reflect the need for only two alignment pin holes instead of the four bolt holes.
And based on Redoxino's presentation at the Nordic Flow Battery Network meeting, Gastronorm (GN) containers are a nice standard to use for secondary containment, it seems with some tweaks we could fit the entire kit into a GN 1/3 150 mm deep container, which are a standard for industrial kitchens worldwide (and available in polymers like PP and PC).
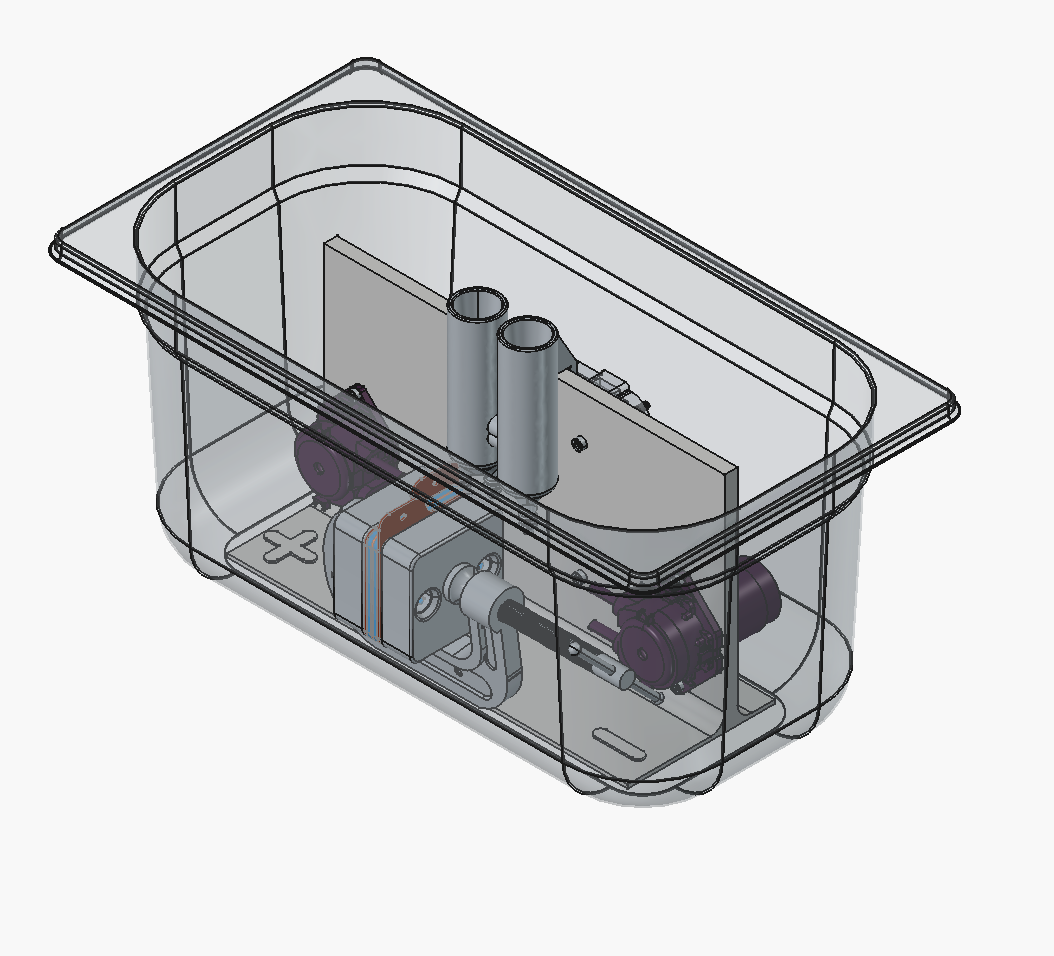

Need to make the reservoirs shorter or mounted lower somehow, and maybe make a holder in the jig for the C-clamp, so that it's in a fixed position.
Right now I'm working on these developments on the
clampable-cellbranch here: https://codeberg.org/FBRC/RFB-dev-kit/src/branch/clampable-cellLet me know if you have feedback/ideas/suggestions! Docs are not yet updated for this configuration, I will do that before I merge it to main.
-
New ion exchange membrane recipe using water softener resin and PVC cementFirst off, @rowow , thanks for making your membrane approach open-source! Out of curiosity, is the patent application alongside it meant to prevent patent trolls from taking advantage of it?
@danielfp248 has looked into membranes quite a bit and I agree that the only way to know for sure about a membrane's chemical compatibility is to test it with the proposed electrolyte during operation, where it will be exposed to, to take our standard zinc-iodide cell as an example, zinc dendrites (which can puncture an IEM) and high concentrations of triiodide, which can be pretty aggressive and "weird", in the sense that it has attacks and goes through many polymers that are otherwise resistant to similar classes of chemicals.
So far we've used paper and Daramic (polyethlyene + silica microporous separators used in lead-acid cells) because our chemistries are meant to be symmetric (and can tolerate mixing) and they are generally quite chemically resistant and handle dendrites better than an IEM. We're not against IEM use by any means, but so far we haven't spent much time on them because of the previously mentioned problems, and it's not our main focus/skill set. We've had our hands full with electrolyte development and cell/system design, so we've essentially opted to keep the membrane "can of worms" (from our perspective) closed---that said, it would be really interesting to test your membrane approach in the dev kit with say, Zn-I, to see how it compares to Daramic---I just don't think we have the time right now to fabricate the membranes ourselves.
It seems you probably have most of the resources already to test your membranes with ZnI? The other thing that would be quite different in terms of membrane requirements for a battery vs. a refining process would be conductivity, I'm not sure if you've done measurements in terms of Ohm*cm² but this would be a harder target to reach for RFB applications.
-
New member introduction thread!Welcome @saphnich and @rowow ! Membranes are definitely something relevant to our work here, to date we have avoided ion-exchange ones like Nafion due to the high cost but having a low-cost and open-source option would be great. I'll hop into the thread @rowow started on DIY membranes.
@rowow said in New member introduction thread!:
Secondly, using foam core PVC sheets which are readily available and cheap from cabinet shops like imeca allows for complex flow cell designs to be easily and rapidly produced with a simple CNC router on various sizes. I have a flow cell design already I'll be glad to upload.
This would be great to see! Feel free to start a thread in @general-discussion about your cell design. We had tossed around the idea of 2D-material milling/laser approaches to flow frames, but have stuck with 3D printed designs for now so that we can have internal geometries in the flow frames - 2D would certainly be easier and cheaper to make, but I was hesitant about the increased gasketing required/adhesives for sealing.
-
Preprint from Forner-Cuenca group: RfbFoam: An open-source framework for porous electrode modeling in redox flow batteriesAvailable here: https://chemrxiv.org/doi/pdf/10.26434/chemrxiv.10001658/v1
Code repo: https://github.com/mbarzegary/RfbFoam
Could be useful for improving the flow field design of the large-format cell.
Great project for someone to take on, without needing to do chemistry or wet-lab experiments

