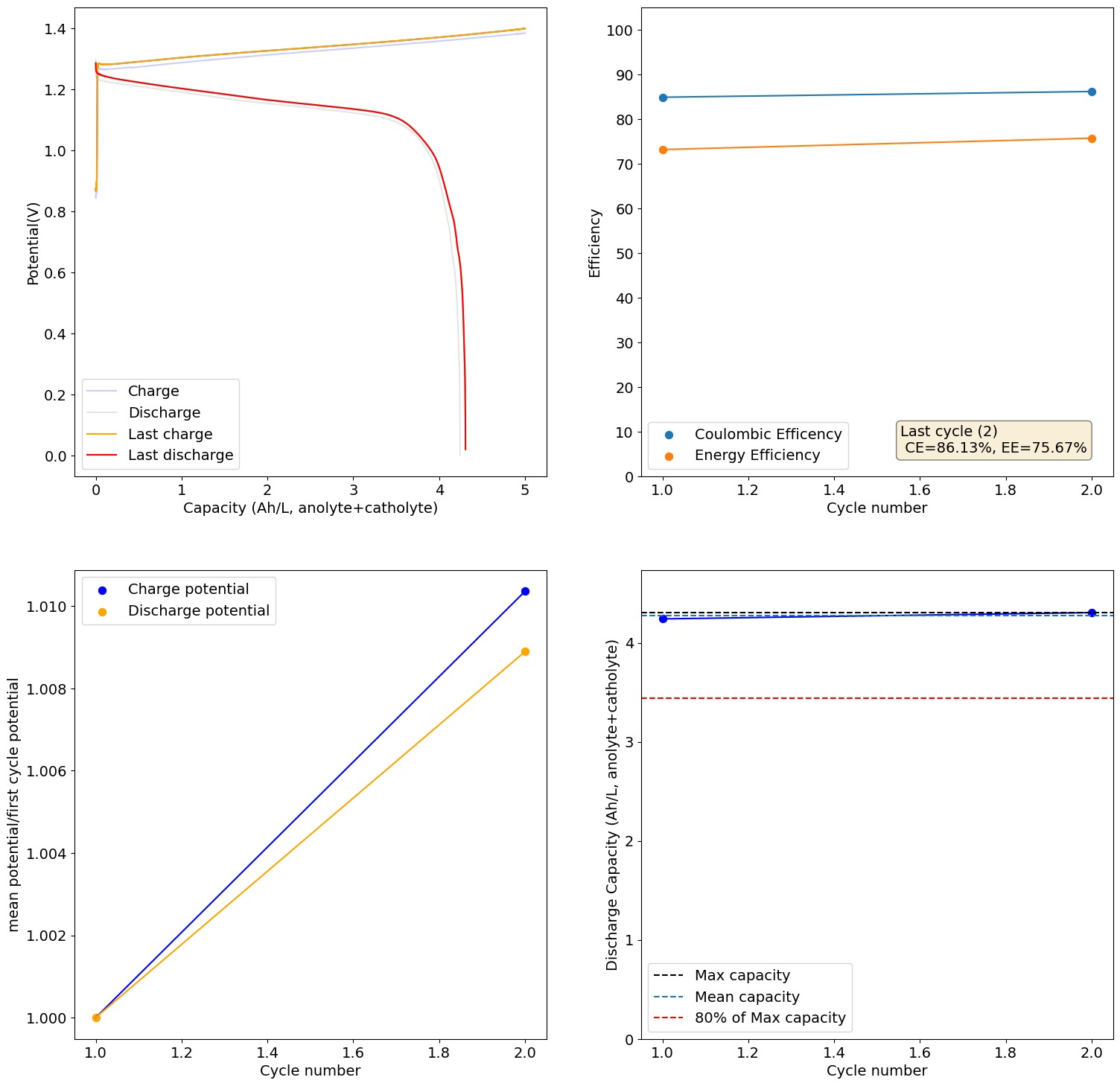
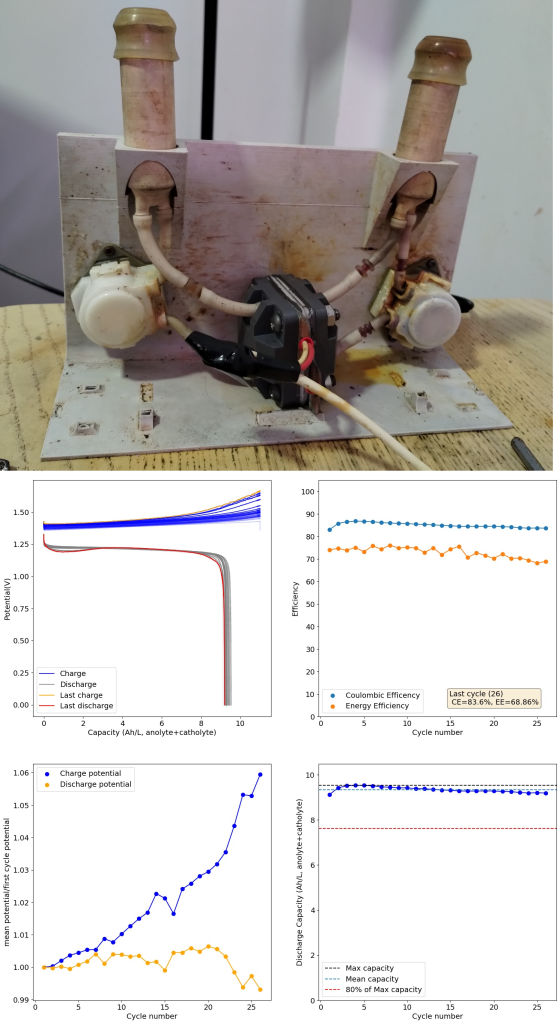
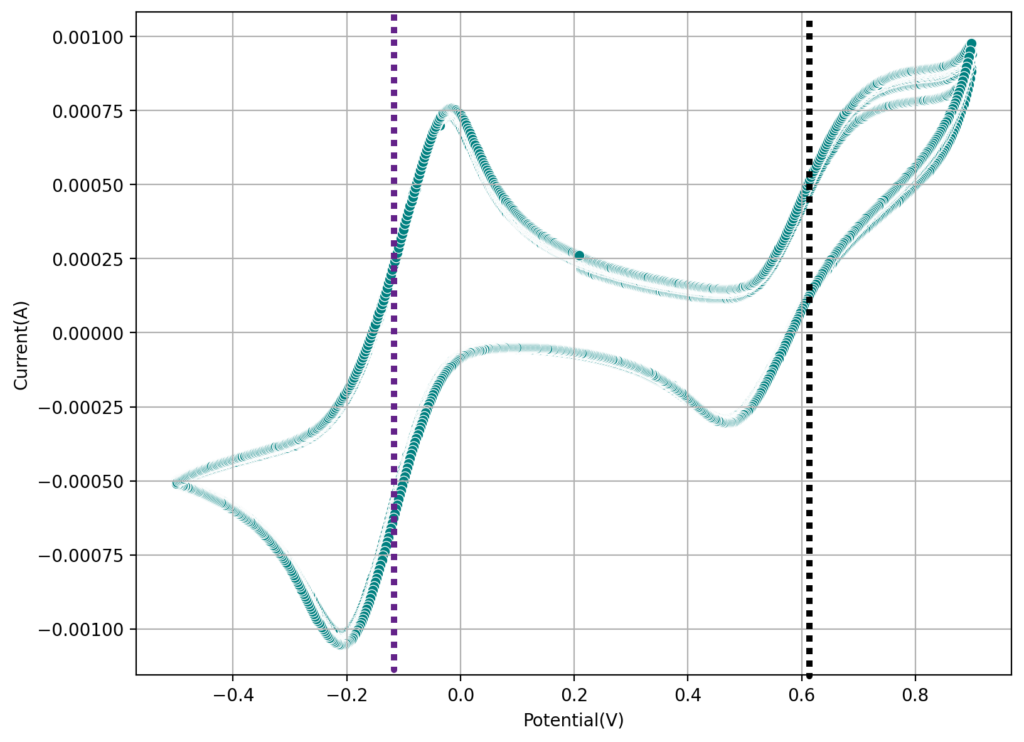
Importing a previous discussion from our old forum:
kirk 1 September 23, 2024, 2:05pm
For example, for residential storage applications:
• 1 kW / 10 kWh
• 70% system roundtrip efficiency including balance-of-plant
• Chemical safety risk no greater than an equivalently-sized lead-acid battery bank
Determining this ahead of time will help us chart an efficient path to get there, and
hearing from real-world potential users will inform our R&D plans!
What applications would you use a flow battery for, and what performance metrics
would you desire?
Sanli 2 September 25, 2024, 6:56am
I think 70% is a good target, but for the mid-term. The existing commercial RFBs also
don’t provide 70% and the low hanging improvements are in the system level (tanks,
pumps, etc) before dealing with stack and electrolyte.
As for the safety, it would be good to look at the existing standards for volume of acid
beyond with a separate compartment is needed, I think it is about 250 litres, but I
might be mistaken.
kirk 3 September 25, 2024, 7:28am
Good point about the secondary containment requirements for the liquid. This
regulation probably differs around the world but would be good to look into. There
are double-walled plastic tanks that count as secondary containment, but these are
probably harder to get. For our first iteration it would make sense to pick a volume
that is more manageable. Putting a big drip tray under the whole setup would be
sensible too.
Dogpoo 4 October 7, 2024, 2:20am
Maybe want to be thinking about insulation, or even the option of a heating element
if potentially users are going to stores the system somewhere like a utility room,
cellar or garage where temperature can vary.
kirk 5 October 13, 2024, 9:02am
I’m not sure at which temperature you’d need insulation. The battery will self-heat to
some degree during operation.
Also, we will likely have an ambient temperature sensor and/or electrolyte temp
sensor in a real system. The concentrated electrolyte should lower the freezing point
of the electrolyte below that of water, but we’d have to do real tests with an
environmental chamber or similar to really understand the viable temperature
window.
Dogpoo 6 October 13, 2024, 9:26am
Good. Etc etc etc and so on and so forth. 20 characters.
julianstirling 7 November 11, 2024, 2:45pm
Secondary containment will be a “fun” thing to try to enforce for the open project as
others start to experiment. I have found in the past that seemingly unneeded things
often get ignored. Lots of caution messages explaining the need for secondary
containment probably go a long way towards this.
kirk 8 November 12, 2024, 8:36am
Yes, good point, we will need to have lots of caution messages all around the
documentation. For R&D purposes, we want people to be able to conduct tests using a minimum amount of materials, but there are always chemical risks no matter the
quantity. For our benchtop system, the volumes are so low (around 10 mL total) that
it shouldn’t be an issue, but for stack testing, we’ll have to spec an option, the more
affordable the more likely people are to use it. And add some images and warnings of
examples of chemical accidents where lack of secondary containment caused issues.
julianstirling 9 November 12, 2024, 9:14am
Yeah. I have taken a quick look through he docs I see there are quite a few.
While it wasn’t safety related, we used to find for OpenFlexure everyone ignored on
the printing page that the optics module should be printed in black. This changed
when we started both explaining why and adding it to the checks:

then when you come to assemble it:

It seems that the short bullet point sentences really helped people not miss what used
to be in longer form text. The information symbol link to more detail.
Dozuki had a really nice presentation about how to do documentation that really
helped me. I’ll see if I can dig it out. Or if not remember the key messages.
pinecone 10 January 27, 2025, 8:15pm
What applications would you use a flow battery for, and what performance metrics would you desire?
I’m thinking about intra-day arbitrage of market-priced electricity. The ratio of
average consumed price to lowest daily price (night) is consistently about 10x or
more here, so there is some potential to save money by time-shifting consumption.
Do you have a cost estimate for a 1 kW / 10 kWh system (like above)? This would be
more than enough to shave off the price peaks for a single household.
kirk 11 January 29, 2025, 11:44pm
Welcome to FBRC, @pinecone !
I don’t have a straight-up answer for you right now. We don’t have a cost model or
estimate yet but we would like to and will have to build one in time. I did some basic
cost modeling in the past but for different chemistries/systems. It wouldn’t be too
hard to have a simple spreadsheet for back-of-the-envelope style calculations.
Daniel posted on another forum some basic calcs for a larger system that someone
had asked about: https://diysolarforum.com/threads/my-adventures-building-a-diy-zn-i-flow-battery.69145/post-873727
My adventures building a DIY Zn/I flow battery | DIY Solar
Power Forum
Quoting him here:
DIYrich said:
What is the usable energy of 30,000 litres?
What is the cost of 30,000 litres?
I’m wondering if it can be used for shifting summer production to winter
usage.
15,000 catholye + 15,000 anolyte at 35Ah/L would give you 525kAh which at a
mean discharge voltage of 1.23V would give you 645 kWh, this is 0.645MWh, so
very massive system. At 1mL per cm2 of electrode area you would also need to
have 1500 m2 of electrode area, which at a standard 25cmx25cm per cell would
imply having at least 24,000 cells. This is a massive system. Probably a couple of
containers filled with stacks of cells to process what is literally a pool of
electrolyte. Since the energy efficiency is 70-75%, you will need to put at least
0.86MWh in to get that 0.645MWh out.
At bulk prices of:
ZnCl2 - 1700 USD/ton
4 of 6
KI - 2900 USD/ton
NH4Cl - 450 USD/ton
For 30,000L you would need 8.17 tons of ZnCl2, 3.20 tons of NH4Cl and 19.9 tons
of KI. The total cost of the salts would be 32.1K USD.
The above doesn’t include pumps, tank costs or cell costs. Note that since no ion
selective membranes are used, this is going to be significantly lower cost
compared with a Vanadium based system. Big systems have significant additional
issues - for example pumping efficiency becomes a huge concern - so I’ll have
clearer costs for you once we implement the first 25x25cm cells.
We are however FAR from anything at this scale. Right now we are focusing on
the small scale. Once everything is optimized the costs for larger scales might also
drop further. Hopefully significant improvements in the energy density are still
possible since the solubility does allow for much higher densities.
Zinc-iodide isn’t the cheapest possible chemistry, but it’s working decently as a
starting point.
Daniel estimated 32.1K USD for 645 kWh of usable energy, so scaling that to 10 kWh
is about $498 in chemical cost. The cost related to the 1 kW power component, the
stack, requires more involved calculations, but there’s nothing particularly expensive
component-wise in the stack (like platinum or gold…)—if you’re not using an
expensive membrane. It’s mostly plastic and graphite (in various forms) with two
copper plates and some tie rods, but the design and control of it is very important,
which is what we’re focusing on now.
Peak-shaving and intra-day arbitrage seem like great opportunities for RFBs though,
that is definitely something we’d like to eventually see happen!
pinecone 12 January 30, 2025, 9:08am
Thanks for the info. The chemicals are surprisingly expensive.
My very rough estimate of the break-even cost for a 10 kWh peak shaving system is
about 1000 EUR, which does not leave much for the rest of the hardware after
chemicals, even when allowing for DIY construction, 3D printed parts etc.
This is a cool project though, best of luck!
kirk 13 January 30, 2025, 10:51pm
Thank you! And yeah, we are trying to develop a functional system, but it won’t be
economically competitive as a DIY build for quantity=1—there would have to be a
group buy or a business set up to buy chemicals in bulk, flow frames injection
molded, etc. This project is for the R&D to get to a viable system, if we get to a
functional system the idea is the project output’s are licensed for commercial use,
and a real business could make it more affordable at scale.
A kit build may be possible if there was a supplier for some of the specialty
components or similar.