Made some Linux AppImages of improved MYSTAT software for anyone who uses it! Also runs on Windows ofc, it's just Python.
World
Topics from outside of this forum. Views and opinions represented here may not reflect those of this forum and its members.
-
Everything about energy production and storage.
Related communities:
A world of content at your fingertips…
Think of this as your global discovery feed. It brings together interesting discussions from across the web and other communities, all in one place.
While you can browse what's trending now, the best way to use this feed is to make it your own. By creating an account, you can follow specific creators and topics to filter out the noise and see only what matters to you.
Ready to dive in? Create an account to start following others, get notified when people reply to you, and save your favorite finds.
Register Login-
Lab Notebook Entry #16
"Finished" the argon Zn-I test, but because it leaked. Made improvements to the dev kit design.
https://dualpower.supply/posts/lab-notebook-16/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
Lab Notebook Entry #15
10 cycles/2 days into the argon Zn-I test. It looks weird but... also good? I probably just jinxed it.
https://dualpower.supply/posts/lab-notebook-15/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
Lab Notebook Entry #14
Designed new reservoirs with Luer Lock fittings, started a Zn-I test under argon (Schlenk technique with welding gas), making progress on battery cycling setup for large-format cell
https://dualpower.supply/posts/lab-notebook-14/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
-
Lab Notebook Entry #13
Made it to 46 cycles/200 hours, the famed Zn-I "voltaic bulge" appears, blaming it on ambient oxygen. Time to bust out the inert gas

https://dualpower.supply/posts/lab-notebook-13/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
@idlestate also, some studies of these systems I think point to complexes being formed in solution, that contain both chloride and iodide/iodine, like ICl-, for example. They might show up with some spectroscopic techniques like Raman? So chloride can play more of a role than just supporting electrolyte. -
Lab Notebook Entry #12
30 cycles, 133 hours, and now a UPS! Which has already saved my butt. And easier-to-read plots.
https://dualpower.supply/posts/lab-notebook-12/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
Lab Notebook Entry #11
Cell cycled most of weekend, power cut interrupted cycling. 25 cycles, 110 hours, seems stable.
https://dualpower.supply/posts/lab-notebook-11/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
Managing electrolyte volume imbalance in flow battery testing / Lab Notebook Entry #10
16 cycles and 72 hours, signs of periodic self-balancing of electrolyte volumes
https://dualpower.supply/posts/lab-notebook-10/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
Filmed myself building a cell so you can see the whole process start-to-finish! Still working on updating the documentation for the clampable-cell branch. It works pretty darn well without any bolts.
-
Starting a thread to keep track of our zinc-iodide development. Background in this blog post: https://fbrc.dev/posts/progress-update-dev-kit/#preliminary-results-from-first-chemistry-zinc-iodide
Current electrolyte composition is roughly 2M KI, 1M ZnCl2, 2M NH4Cl, 5% triethylene glycol, with a Daramic separator and graphite felt on both sides, no spacer.
-
Hi folks,
Over a couple weekends, I've been working on parameterizing the FBRC model. It was borne of a couple kinks I ran into - I would want to adjust bolt or hose barb diameters based on what I had lying around, or scale the model down for smaller lab scale setups while maintaining control over electrode thickness for lab experiments. The idea is to adjust a single parameter in a script, which then adjusts the appropriate dimension in all the design files, then spits them out for fabrication.
The result is a single "model" with flexibility to adapt plumbing, sizing, and construction according to your particular application. So for example, the same parameterized model can spit out
a small cell with hose barbs for lab scale applications (18cm2 electrode area)

a medium size cell (using the current FBRC large format sizing)

or a "fat stack" (3500cm^2) with extra, thicker bolts

all from the same parameterized design. Once the designs are generated, all the associated STL, STEP, and DXF files can be downloaded for fabrication.
If this is of any interest to you, I'm hosting the parameterized model in a webapp here and I've uploaded a short video walking through the general process here.
I briefly discuss it in the video, but because the models are parameterized, it's easier to programmatically keep track of the locations of ports, walls, manifolds, etc. That makes boundary condition declaration and the like much easier to automate as well - so I'm in the process of slotting this into an automated hydraulics/electrochemical simulation suite. Imagine generating your custom model and simulating a "digital twin" for estimates of performance prior to purchasing materials or finalizing design.
I'm very open to constructive criticism on any part of the project (from ideation to UX to engineering). Have fun fiddling with it and let me know what you think!
Also, this reminds me of the OpenAFPM project - they also use FreeCAD and provide a dashboard for people to input custom parameters for a small wind turbine generator, there is some FEM, and then design files are output. There is a video demo here: https://www.openafpm.net/cad-visualization I know a few of the folks behind that project, I'm sure they'd be happy to give input on how to accomplish a similar goal but for flow batteries. -
Lab Notebook Entry #9
Finally a decent test cell underway (it's not done yet!)
Error bars getting smaller, energy efficiency over 70% for our standard zinc-iodide system.
https://dualpower.supply/posts/lab-notebook-9/
#academia #OpenScience #Quarto #batteries #EnergyStorage #energy #science #electrochemistry
-
-
This guy did some videos for an all iron static battery. Here is the final version video and info page:
https://www.hardware-x.com/article/S2468-0672(25)00007-0/fulltext
The cost per kWh of this is too high, the use of organic materials and ion exchange membranes puts this closer to what people do in academia and further from the cells that can be reasonably fabricated using DIY approaches. The use of an ion exchange membrane likely reduces the lifetime of this cell a lot. For a static cell, I think the sulfuric acid Cu/Mn battery we have discussed before is far more promising. That chemistry requires no exchange membranes, uses only commodity inorganic materials and cycles to 30-40Wh/L. For pure Fe batteries I am much more inclined to the Fe flow batteries. -
There's a lot I would love to add to this community. I think the way traditional methodology for this technology needs to be reapproached. Firstly, by using my open sourced membrane recipe you can glue it directly into a PVC cell. Secondly, using foam core PVC sheets which are readily available and cheap from cabinet shops like imeca allows for complex flow cell designs to be easily and rapidly produced with a simple CNC router on various sizes. I have a flow cell design already I'll be glad to upload. I'm new here so sorry there's a lot I love to want to share and am doing things one thing at a time.
You can find more details on the membrane on the following GitHub https://github.com/Rowow1/Open-sourced-off-the-shelf-ion-exchange-membrane
However I started a separate thread specifically for this membrane topic.@kirk said in New member introduction thread!:
This would be great to see! Feel free to start a thread in @general-discussion about your cell design. We had tossed around the idea of 2D-material milling/laser approaches to flow frames, but have stuck with 3D printed designs for now so that we can have internal geometries in the flow frames - 2D would certainly be easier and cheaper to make, but I was hesitant about the increased gasketing required/adhesives for sealing.
The key aspect of using the proposed PVC membrane recipe together with the pvc foam board actually completely eliminates the need for gaskets. You simply glue the membrane in place since its also made from pvc. Overall there's far less labor and requirements, forming a sealed (technically welded) bonded cell for a fraction of the price that 3d printing would cost. The long term goal and my personal next goal is to get into injection molding which of course is the best option.
Ill see about uploading the cell fusion 360 model soon depending on if people even care or not. But the point is the core principle idea of being able to use much cheaper pvc foam board material and mass produce these cells at a much better scale than one layer at a time with 3d printing. I was able to make unique geometries needed for flow cells with my 2d design.
As a comparison, you can buy these foam boards for $60-80 for 1/2" or 3/4" from imeca for a full 4x8ft sheet
@rowow said in Using cnc router cut PVC foam boards as cell frame: The long term goal and my personal next goal is to get into injection molding which of course is the best option. At scale, definitely! @rowow said in Using cnc router cut PVC foam boards as cell frame: Ill see about uploading the cell fusion 360 model A .step file would also be great so I could look at it in FreeCAD! I don't have a Fusion 360 license. I'd be curious to see if we could take a similar approach. -
Hi all, been planning some improvements to the dev kit that are underway, based on some ideas exchanged at a conference with some other open-source flow battery projects (Redoxino and the team at QUB).
It's mostly to improve ergonomics - there is now a case to hold the arduino and wires on the rear of the cell:
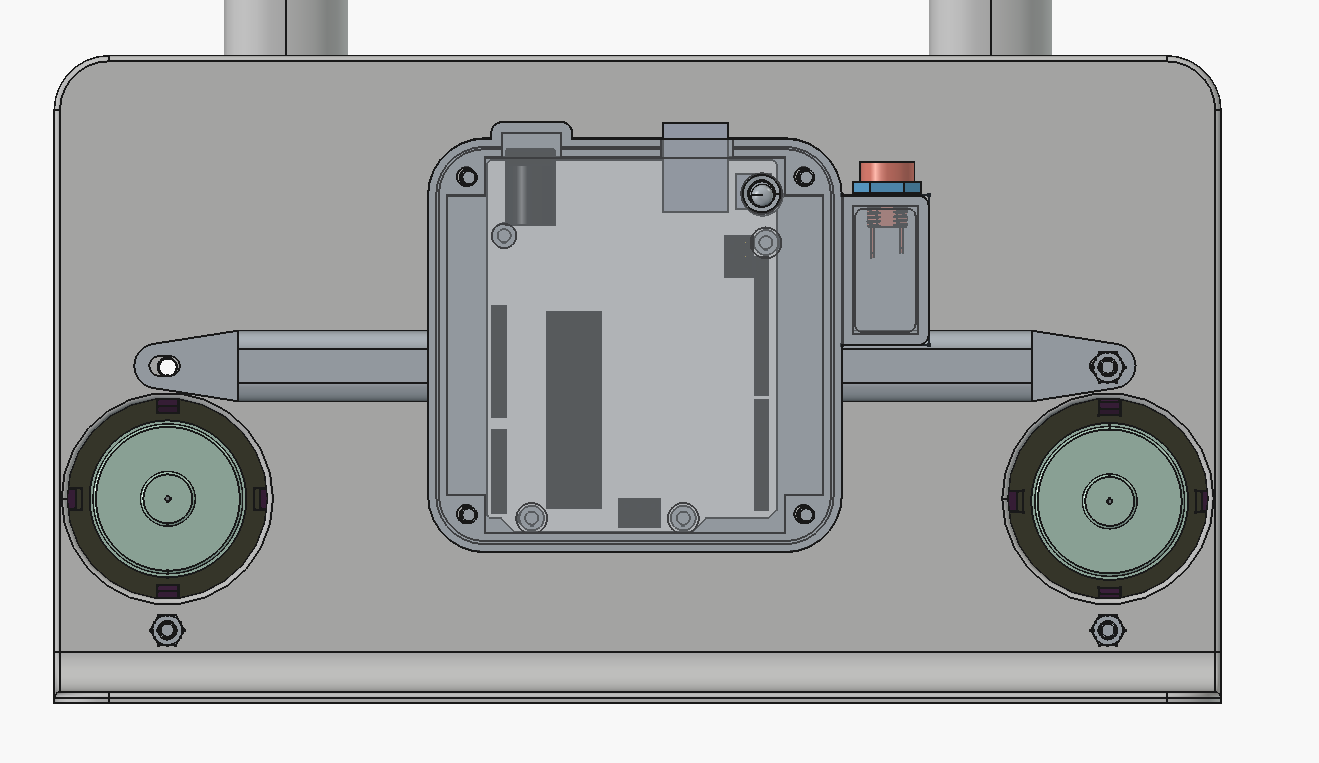
We also realized you can just clamp the cell shut with a 2-inch C-clamp instead of using the bolts, and it seals well:

Note, the new endplates here used are backwards-compatible with previous gaskets/graphite plates/brass current collectors, so no need to recut anything. Moving forward though, the design files for those components will change to reflect the need for only two alignment pin holes instead of the four bolt holes.
And based on Redoxino's presentation at the Nordic Flow Battery Network meeting, Gastronorm (GN) containers are a nice standard to use for secondary containment, it seems with some tweaks we could fit the entire kit into a GN 1/3 150 mm deep container, which are a standard for industrial kitchens worldwide (and available in polymers like PP and PC).
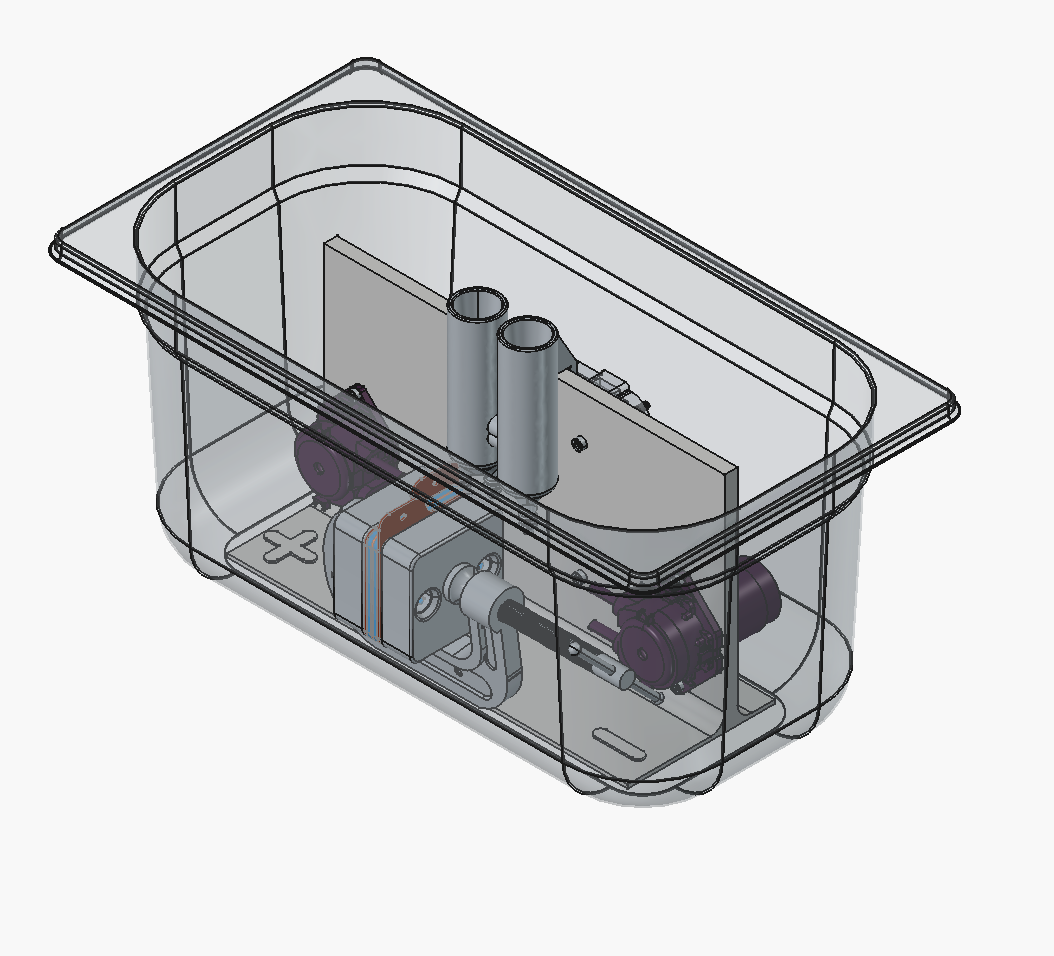

Need to make the reservoirs shorter or mounted lower somehow, and maybe make a holder in the jig for the C-clamp, so that it's in a fixed position.
Right now I'm working on these developments on the
clampable-cellbranch here: https://codeberg.org/FBRC/RFB-dev-kit/src/branch/clampable-cellLet me know if you have feedback/ideas/suggestions! Docs are not yet updated for this configuration, I will do that before I merge it to main.
@doho said in Upcoming improvements to the dev kit: I have try-ed to download Your new files from github (.stl and .pdf), but the files all appeared as broken, even in github. The files to sprint for the new endplates are here: https://codeberg.org/FBRC/RFB-dev-kit/src/branch/clampable-cell/CAD/exports [image: 1771858819246-1c46bc80-7e99-4780-8178-46d24a289a32-image.png] I haven't updated the new You have to download them individually, unless you clone the repository with Git LFS set up---the large CAD files are handled with LFS and without LFS installed downloading the repository just downlaods a pointer/reference to the CAD file, not the actual file (at least that's how I understand it). Let me know if this works for you! -
My individual changed construction
/FBRC-Redox-FLOW-Batterie/Fotos Projekt/IMG_7768.JPG
As I had to change the tubes in my pumps (they were the wrong tube type),(thanks @sepi) I also changed the connectors to Luer Lock .
Therefore it was necessary to enlarge the boreholes in the endplates and in the brass-plates to 10.5 mm to get the female Luer Lock part through.
And I changed my construction to sucking from the cells.Some other thoughts:
@danielfp248 In your picture of 08.01.26 you put the pump for suction on the lower side of the cell, why?
In my opinion it would be better to connect the pump upper side to avoid air in the system as I did in my setup.@kirk
your new approach for the test cell seems veri interesting. But for getting reproducible resultants the flow through the cell should be down to up, but not horizontal (how much would be air isolating in the chamber when changing parts?)I do not have an idea for a reasonable physical layout for a really simple change of the test-chamber, but I will think over.
Printing materials I have used:
Parts not in contact with chemicals: PETG, works very well
PP: My 1st: (Yousu),all surfaces o.k, but all barbed broke. 2nd: Innovatefil PP GF worked very well, but the upper surface was rough, tests will come.
Very nice work @doho ! It's great to see your setup. @doho said in My Suction Luer Lock: But for getting reproducible resultants the flow through the cell should be down to up, but not horizontal (how much would be air isolating in the chamber when changing parts?) This is a good point, from what I've seen in other applications cells should generally clear air/produced gases by flowing against gravity from bottom to top, that is still possible with the new setup but the tubing from the pump outlet to cell inlet would have to be slightly longer. In my jig redesign I'll take this into account when writing the documentation. -
I just wrote a blog post sharing my first results testing the Zn-Br chemistry recently published by a Chinese group. Sodium sulfamate is not very easy to get, but I was able to source it from labdiscounter.nl in the EU. If any of you are in labs where you can also test this chemistry, I would be happy to hear about your thoughts and/or results.
-
A recent Chinese Nature paper showed how Sodium sulfamate can be used in Zn-Br batteries to sequester active Br2 into an N-bromosulfamate that is much less aggressive, much more water soluble and even more easily electrochemically reversible than elemental bromine. I also wrote a recent post discussing the potential use of nicotinamide to achieve this (plot twist, it doesn’t work as the nicotinamide Zn complex is not very soluble). In today’s post I want to share with you my attempts at reproducing this chemistry of the Chinese paper using our open source flow battery dev kit.
The paper is very extensive and shares multiple formulations, they share a formulation for normal asymmetric cells as well as formulations to run the batteries using microporous Daramic membranes. Thankfully I have a bunch of 900um Daramic (thanks a lot to Daramic who donated these membranes to us for research). I bought some Sodium sulfamate (NaSA), Zinc bromide (ZnBr2), Potassium bromide (KBr) and potassium acetate (KAc) and proceeded to run some tests.

Tests using 1M ZnBr2, 1M KBr, 2M KAc, 1M NaSA. Charge and discharge were both done at 30mA/cm2. My tests using the formulations that they disclose exclusively for daramic were not very successful. Formulations using only ZnBr2, KAc and NaSA suffer from either lower capacities because of lower conductivity or issues with hydrogen evolution. This was specially the case when I tried the ZnBr2 1M, KAc 1.5M, NaSA1.5M formulation, which they suggest in the supporting information to reach >50Ah/L. However I think this is a typo and they meant 2M ZnBr2. If you read that paragraph in the supporting information closely you’ll realize why this is the case (they previously refer to a ZnBr2 1M solution and then say this is basically 2x that, but still write it as ZnBr2 1M).
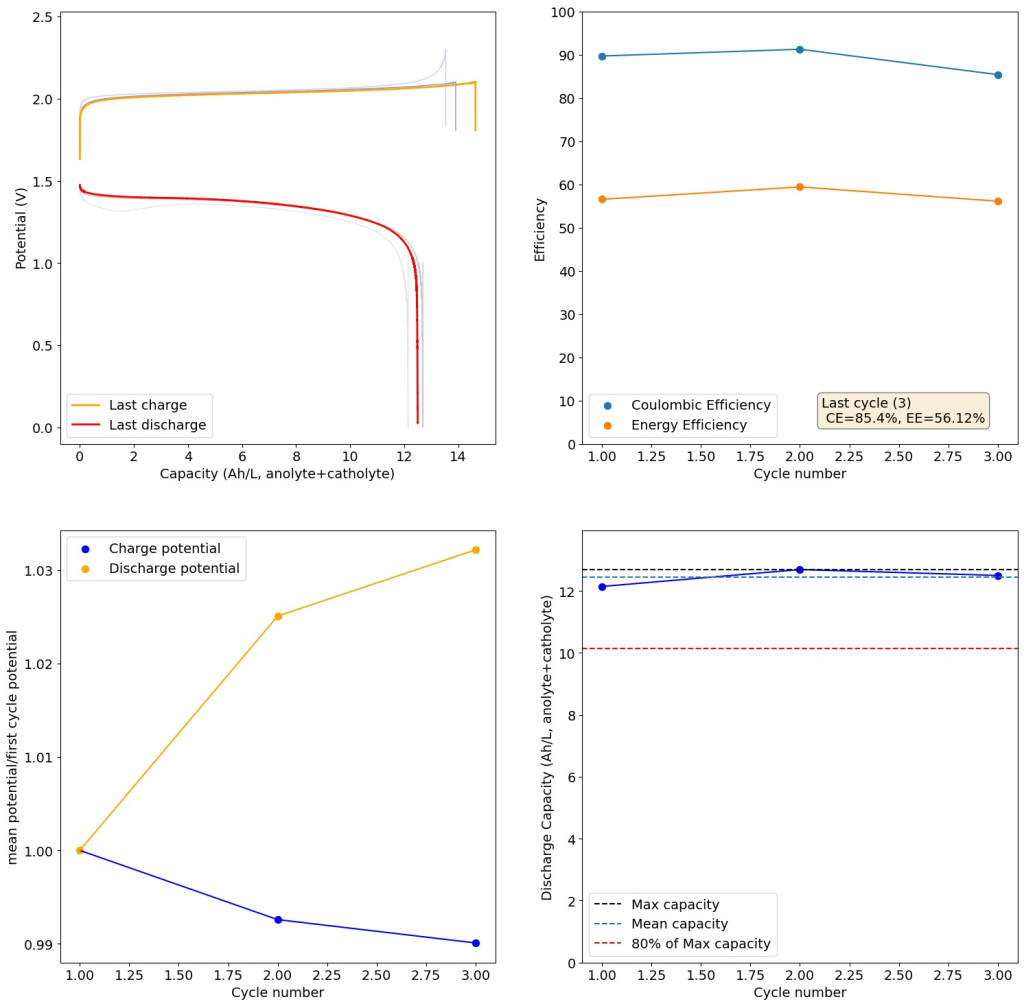
I then proceeded to test using some of the electrolytes they suggest for asymmetric cells, which were much more successful. In particular the 1M ZnBr2, 2M KAc, 1M KBr and 1M NaSA was great, with high CE values and decent EE values (see graph above). I didn’t experience dendrites before reaching the Nernst limit of the cells when using the 900um thick Daramic, which suggests plating is not as aggressively dendritic as with other electrolytes. However dendrites are quite evident when using 300um Daramic, suggesting you need around 300um of Daramic for every 30mAh/cm2. This might explain why the paper restricts most plating to below 90mAh/cm2 when using the 900um Daramic. I have yet to reproduce the Chinese group capacity or cycling stability values, but I believe I have validated the electrochemical principles well.

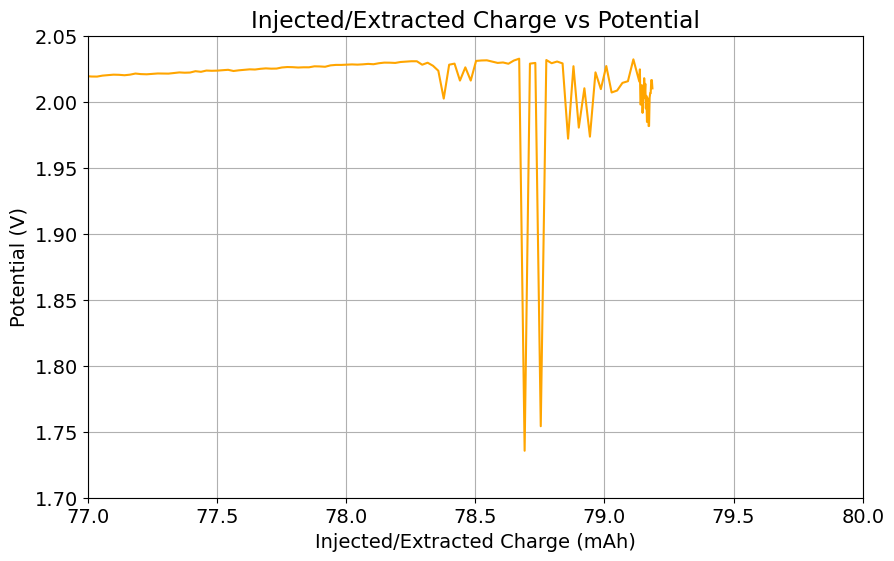
Clear evidence of dendrites at 78-79mAh (using 8mL of total electrolyte) using a 2M ZnBr2, 1.5M KAc, 1.5M NaSA electrolyte. It is also worth noting that the Chinese group does some fancy functionalization of their felt with both nitrogen containing groups and carbon nanotubes, which aggressively boosts the conductivity and energy efficiency of the felt for the Br reactions. This is an important different that might justify why they get energy efficiencies closer to 75-80% while mine are just shy of 60%. I also haven’t optimized the compression ratio of my felt, which means that my felt might be under or over-compressed to extract the max EE in this setup. I also lack an oven to properly do air activation of the felt, so my felt is quite suboptimal and just used as-is.
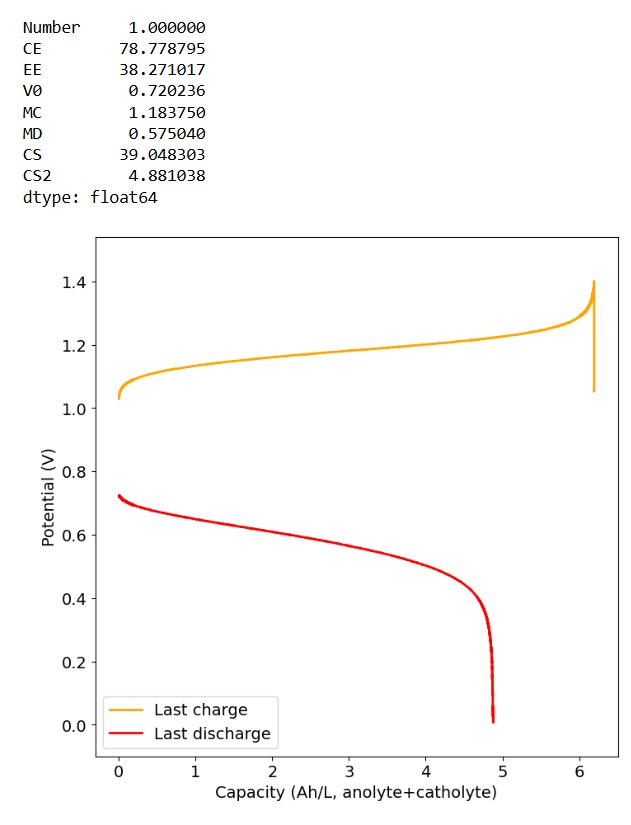
Furthermore, the paper successfully tested a true flow battery setup using Ti-Br. I cannot easily buy TiOSO4 but I decided to try to innovate and test this chemistry in a fully symmetric setup coupled with 0.5M of Fe-DTPA. While Fe-DTPA isn’t expected to be fully resistant to N-bromosulfamate, I figured it might last enough to provide me with some data. Given that the redox potential of the Fe-DTPA redox couple is quite lower than Fe2+/Fe3+, I figured it should give some appreciable voltage in an Fe-DTPA/N-Br-sulfamate battery. Fe-DTPA is also quite soluble and stable at the near neutral pH that favors the N-Br-sulfamate chemistry, so it should work nicely.

Fe-DTPA 0.5M, NaSA 1M, KBr 1M, KAc 1M. Cycled at a current of 30mA/cm2. The results above, which have never been published before, show that this chemistry works to some extent. The low CE does suggest that a significant portion of the Fe-DTPA is somehow lost, perhaps to oxidation by atmospheric oxygen (I cannot purge my cells with N2 or Argon at the moment), but also likely from just interactions with N-Bromosulfamate across the microporous membrane. With that said, it does show that the new stabilized bromosulfamate chemistry opens up the window to some very interesting options that just didn’t exist before. Perhaps I can test nicotinamide in this setup, where there is no Zn to cause it to precipitate out of solution.
Finally, I wanted to dedicate the above post to Robert Murray-Smith, a fellow chemist in the UK who passed away recently and was a key inspiration for the start of this blog. I know his passing has been very sad for a lot of us in the DIY community, the curiosity and inspiration he instilled in a lot of us will live on. Thank you Robert!