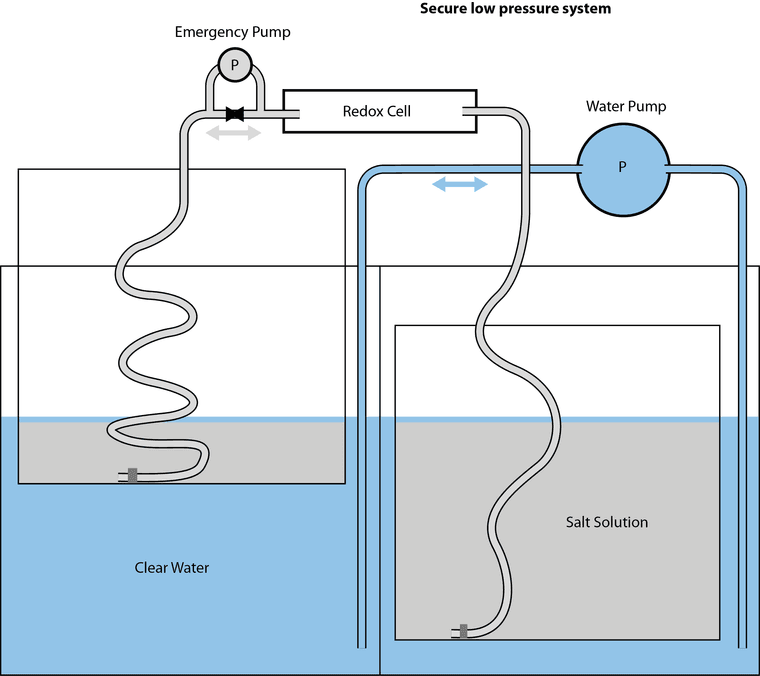
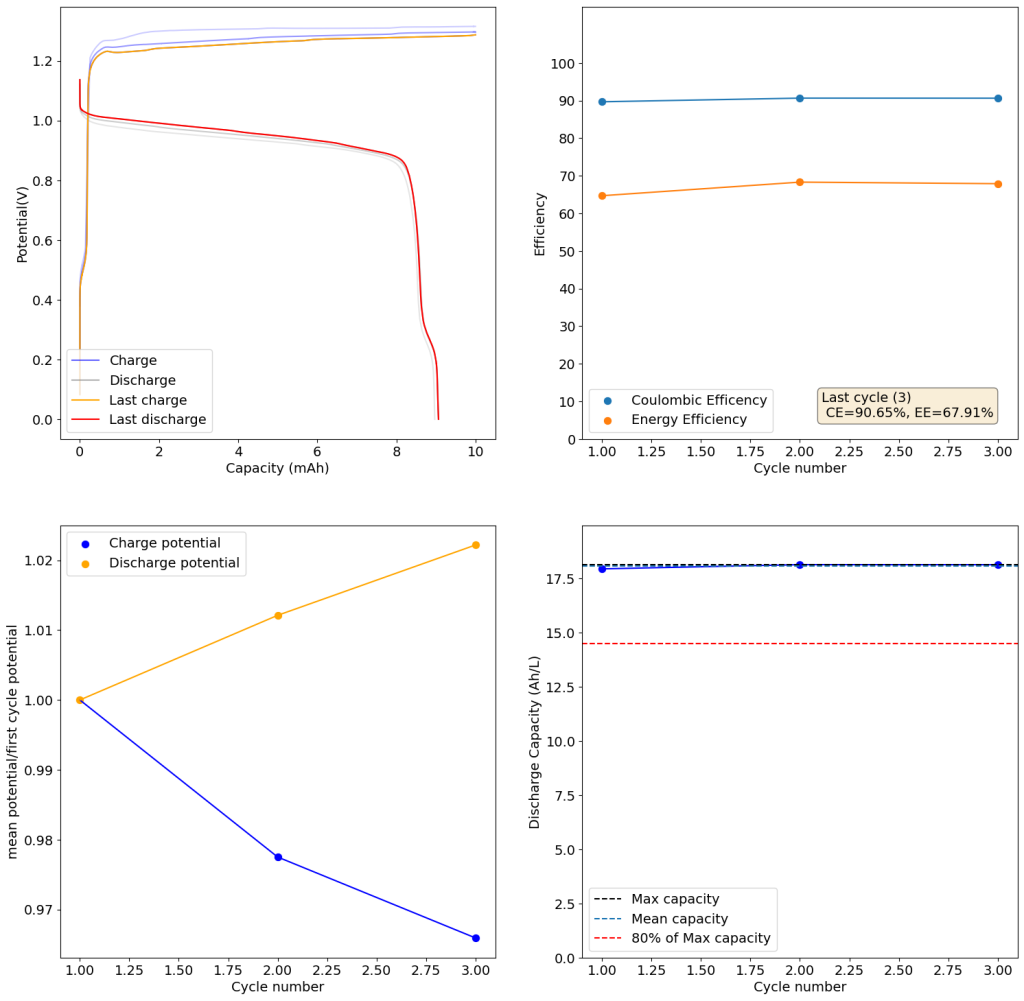
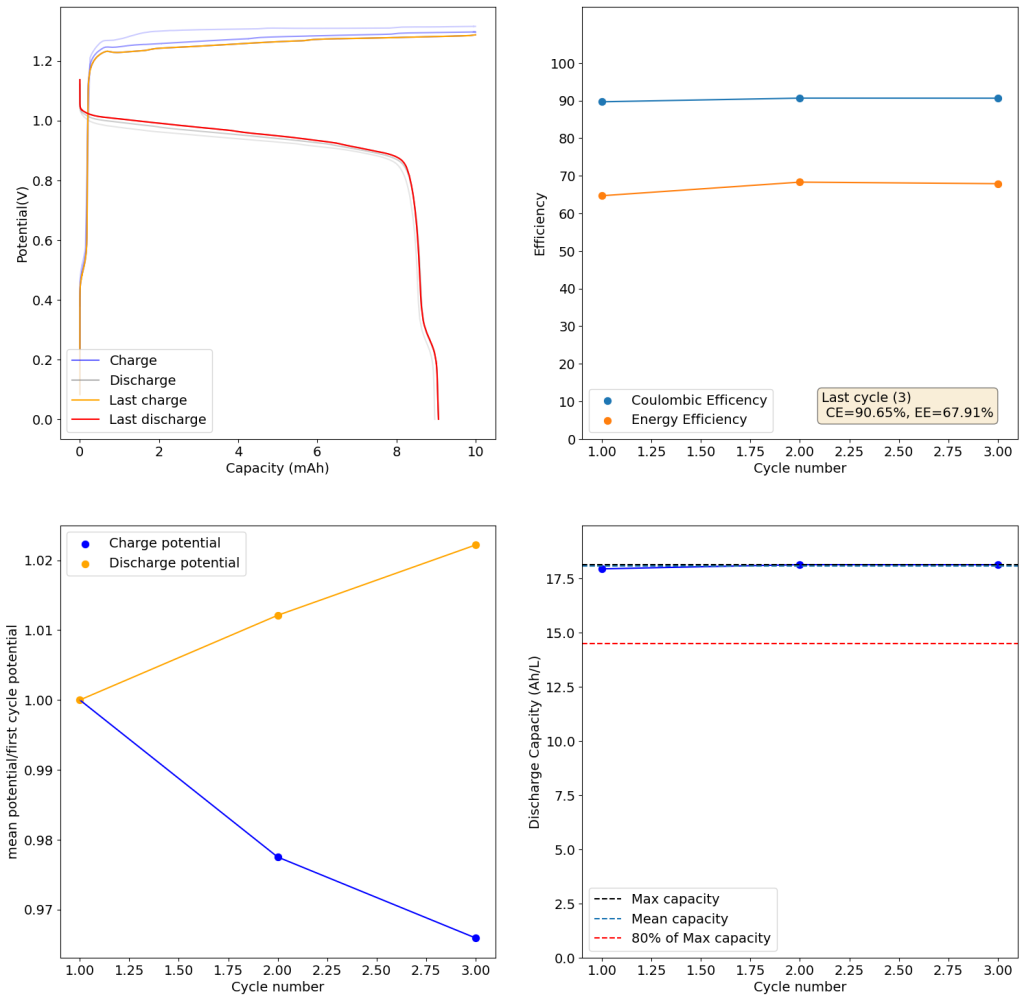
@kirk So my thesis is specifically on slurry/suspension electrodes (instead of using a graphite felt/porous electrode, you suspend conductive carbons with the electrolyte - which also allow you to run solid-phase chemistries in flow) in a way that's sort of chemistry-agnostic. Basically applying chemical reactor design principles to designing slurry electrodes. But here are some salient idiosyncrasies of all-iron cells:
- iron plating in porous electrodes is annoying (acidic = HER, basic = whole host of nasty iron oxides, many of which are quite stable. Plating on carbon substrates is also a pain - most studies I've seen plate onto copper. Also volume expansion is a big pain in static cells. In flow batteries, any time you have plating you end up re-tying power density and energy density through that half cell. Iron plating kinetics are also quite slow, especially in relation to zinc plating.
- Bunch of folks (Savinell and Wainright groups) at case western used slurry electrodes and plated iron onto the slurry particles (ostensibly). They attempted to scale but really struggled with having a performant enough slurry electrode that wasn't too viscous. But plating on suspended particles re-de-couples power and energy density.
- Have you guys looked into water-in-salt electrolytes? They involve dissolving a ton of a supporting electrolyte to the point where they lower the activity of water and suppress HER. I've seen some work using acetate salts and this one using magnesium chloride to support even iron plating - and I've replicated it successfully. The study plates on copper like I mentioned earlier, but I also got it to plate on grafoil.
Solving that performance/viscosity tradeoff in slurry electrodes is part of my thesis! And so is improving the power density of otherwise crappy flow battery chemistries. I'm still working on getting some of it out, but I'd love to share soon or help in any other way.
Material choice from the perspective of mineral centralization and cost is another problem I think regularly about - I did a little study collecting the centralization/governance of various metals and ranked aqueous battery chemistries by cost and "criticality". If that's of interest let me know too!


 The right one was the first, printed with 15% infill, the left one with 60%, but still just 2 wall loops. Of course, you can feel the difference in the weight.
The right one was the first, printed with 15% infill, the left one with 60%, but still just 2 wall loops. Of course, you can feel the difference in the weight.